 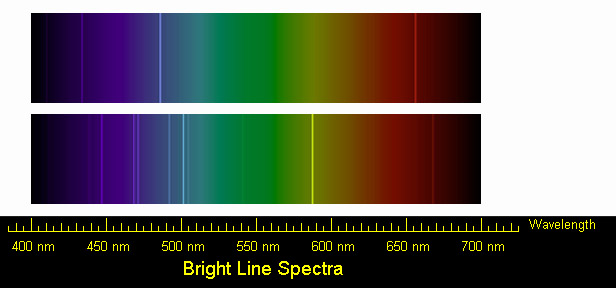
Other series of lines appear in the emission spectrum only and not in the absorption spectrum, and were named the sharp series and the diffuse series based on the appearance of the lines. The term principal came about because this series of lines is observed both in absorption and emission for alkali metal vapours. v Line codes indicate additional descriptive information about the appearance of the spectral line. In either case the in-tensity cannot be assumed to be entirely due to the transition indicated in the classication. The wavelengths corresponding to several transitions are shown. measured spectral line either is blended with another line or has two identications. Figure 10.57 Valence shell energy level diagram for sodium. When electrons descend in energy they produce an emission spectrum. Figure 10.57 shows a portion of the energy level diagram for sodium, which consists of a series of discrete lines at wavelengths corresponding to the difference in energy between two atomic orbitals. 
Fine structure is produced when an atom emits light in making the transition from one energy state to another. The lines are absorption lines when the electron gains energy from an s subshell to a p subshell. fine structure, in spectroscopy, the splitting of the main spectral lines of an atom into two or more components, each representing a slightly different wavelength. The principal series is due to the 3s-np transitions shown here in red. The principal series has given the letter p to the p atomic orbital and subshell. If this is the case, surely it should be a triplet as there are 3p orbitals so each transition would leave a distinct line on the spectrum electrons spectroscopy Share. Helium wavelengths (nm) 438.793 w: 443.755 w: 447. My teacher said that the doublet seen in the sodium emission spectrum is evidence of subshells within the atom. The 589nm \yellow' line of sodium actually consists of two distinct lines, separated by a few tenths of a nanometer. In the next part of the lab you will use your calibrated Michelson interferometer to measure a small di erence in wavelength between two closely spaced spectral lines of a sodium lamp. At the right of the image are the spectral lines through a 600 line/mm diffraction grating. SPECTRAL LINES VIA MICHELSON INTERFEROMETERY. These lines are usually found in the visible and ultraviolet portions of the electromagnetic spectrum. Sodium: At left is a helium spectral tube excited by means of a 5000 volt transformer. In atomic emission spectroscopy, the principal series is a series of spectral lines caused when electrons move between p orbitals of an atom and the lowest available s orbital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
